If, in a hydrogen atom, radius of n^th Bohr orbit is rn , frequency of revolution of electron in n^th orbit if fn , and area enclosed by the nth orbit is

The frequency of revolution of an electron in nth orbit is `f_(n)`. If the electron makes a tran... - YouTube

What is the frequency of revolution of electron present in 2nd Bohr's orbit of H-atom.a)1.016 x - Brainly.in

Fundamental resonance plasma frequency vs electron concentration in the... | Download Scientific Diagram
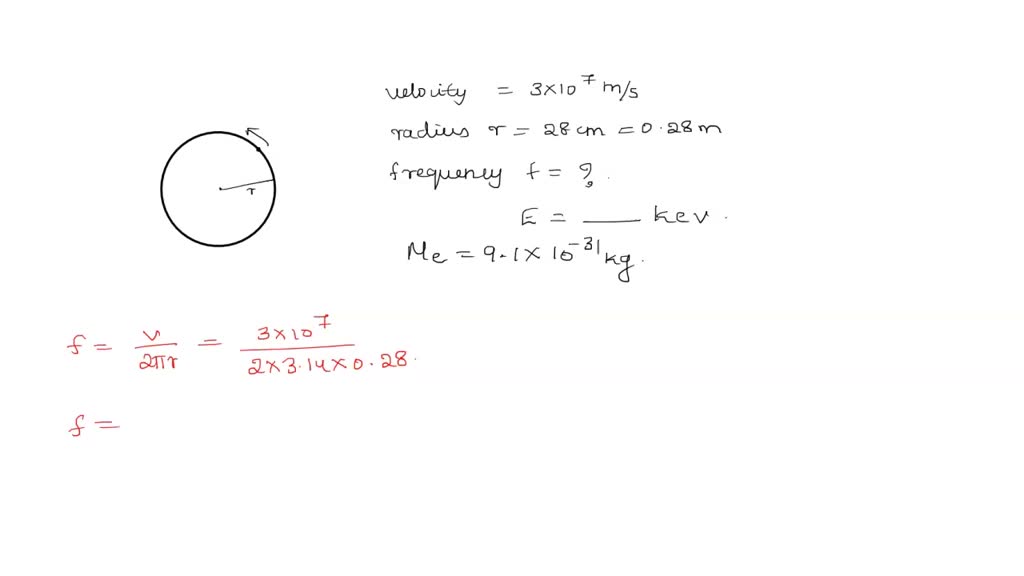
SOLVED: What is the frequency of electron which is moving with a velocity 3*10^7 m/s in the path of radius 28cm. Also calculate the energy of electron in KeV. Mass of electron=9.1*10^-31

Class 12 Physics | #7 Revolution Frequency and Time Period of Electron in nth Orbit in Bohr Model - YouTube

How to Calculate the Photon Frequency Absorbed or Emitted by an Electron in a Hydrogen Atom | Physics | Study.com
Demonstrate that the frequency ω of a photon emerging when an electron jumps between neighbouring circular orbits - Sarthaks eConnect | Largest Online Education Community

Calculate the angular frequency of revolution of an electron occupying the second Bohr orbit of He^(+) ion.

SOLVED: Using the Bohr model of the Hydrogen atom, what is the frequency of the photon released when an electron drops from the n = 4 level to the n = 2
36.Calculate the ratio of frequency of electron in an orbit for 2nd shell of Li+2 to 3rd shell of He+ ion?

Calculate frequency of revolution of electron in 4th Bohr orbit `Be^(+3)` ion . Given that `(pi^(2 - YouTube
Obtain an expression for the frequency of radiation emitted when a hydrogen atom deexcites from level n to level (n−1). - Sarthaks eConnect | Largest Online Education Community



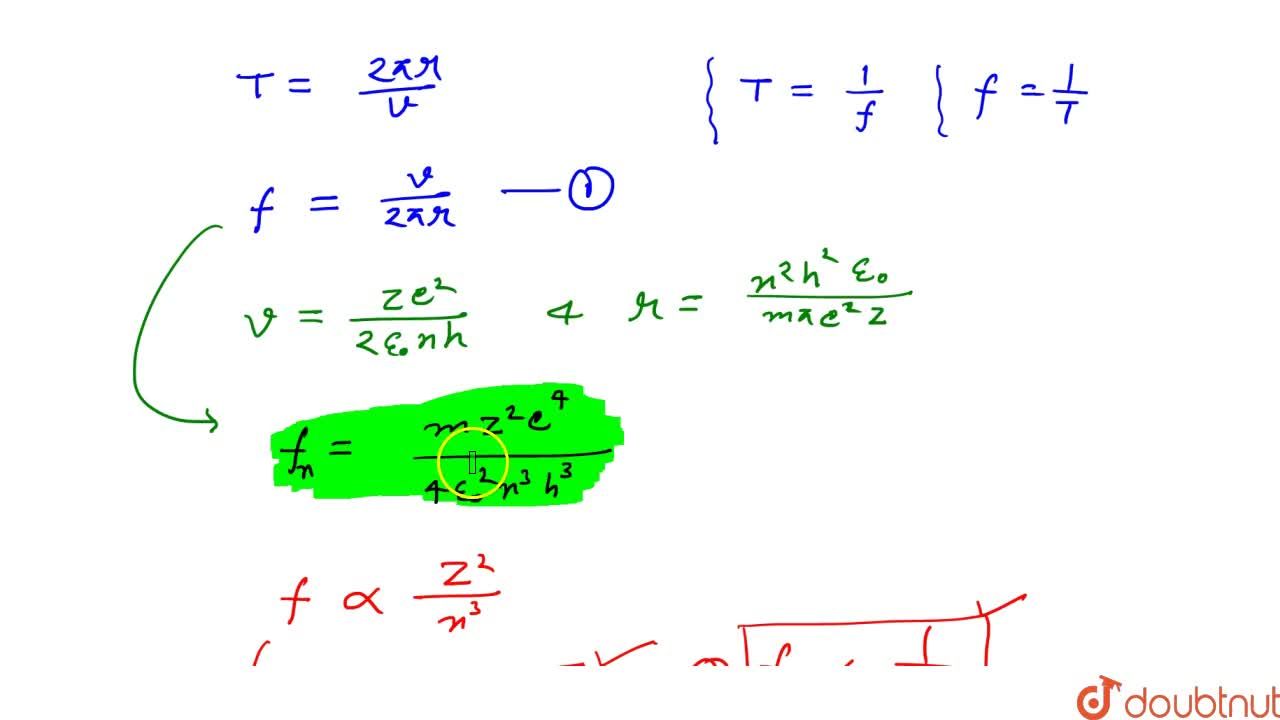

![Term 2] Derive an expression for frequency of radiation emitted when Term 2] Derive an expression for frequency of radiation emitted when](https://d1avenlh0i1xmr.cloudfront.net/135dbe2d-5958-4284-b9a2-e76dd43fdf02/slide1.jpg)